
Digital Medical Education
I am interested in the pivotal role of science communication and digital education approaches in medical education. I am part of the School of Medicine’s Digital Education e-lab, hosted by the HIVE (Hybrid and Interactive Learning Environments). This group is dedicated to pushing the boundaries of innovative pedagogic practice. Our research activity focuses on evidence-based practice that drives teaching excellence. Here, I focus on inclusivity (especially regarding neurodiversity) for both staff and students regarding teaching and how to develop ECRs’ teaching skills.
I am also interested in creativity in medical education, in various ways (crafts, video, illustration), and medical students’ mental health.
Highlighted publications:
– Hassoulas, A., et al. Developing a personalised, evidence-based and inclusive (PEBIL) model of blended learning: a cross-sectional survey. Education and Information Technologies, (2023)
– (Pre-print) Srinivasan, S., et al. Challenging Pedagogical Styles of Teaching: Amalgamating Blended Learning with Near-peer Teaching for Integrated Structured Clinical Examination (ISCE) Preparation.
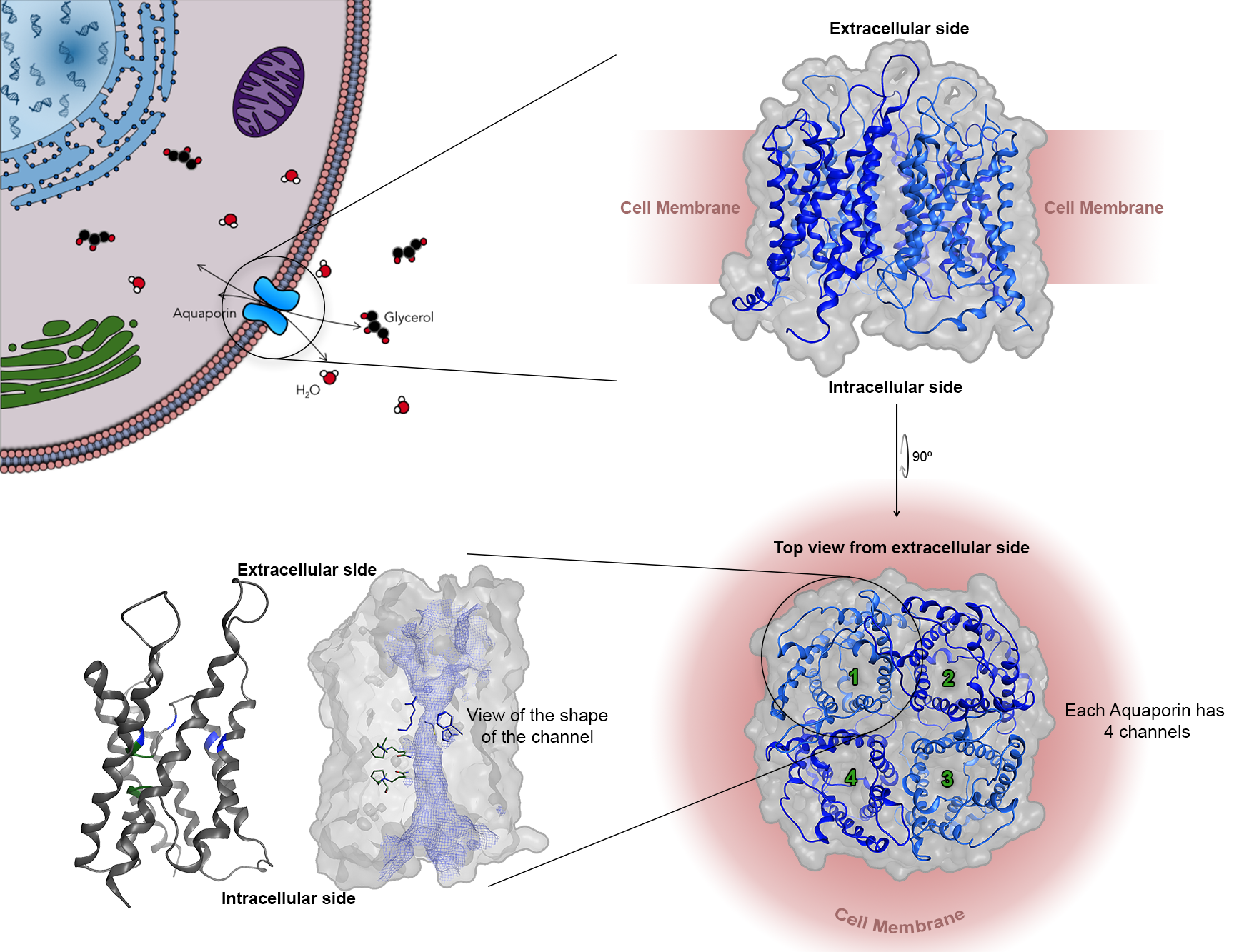
Aquaporins in cancer development
My research focuses on identifying the role of small transmembrane water and glycerol channels, named Aquaglyceroporins, in cancer development. For this, I use known inhibitors rather than genetically altering cells to determine their function and effects when inhibited. I aim to map the function of glycerol and hydrogen peroxide, via glycerol channels, in cancer development, cell migration, differentiation and metabolism. I am particularly interested in how the tumour environment affects the function, expression and localisation of glycerol channels and how their expression, in turn, affects cancer progression.
Currently, I am focused on understanding the Spatio-temporal patterns of aquaglyceroporin expression in prostate cancer under hypoxia and how this contributes to prostate cancer metastasis.
Highlighted publications:
– de Almeida, A., et al. Aquaglyceroporin-3’s Expression and Cellular Localization Is Differentially Modulated by Hypoxia in Prostate Cancer Cell Lines. Cells, (2020)
– Wragg, D., et al. Unveiling the mechanisms of aquaglyceroporin-3 water and glycerol permeation by metadynamics, Chem.: Eur. J, (2019)
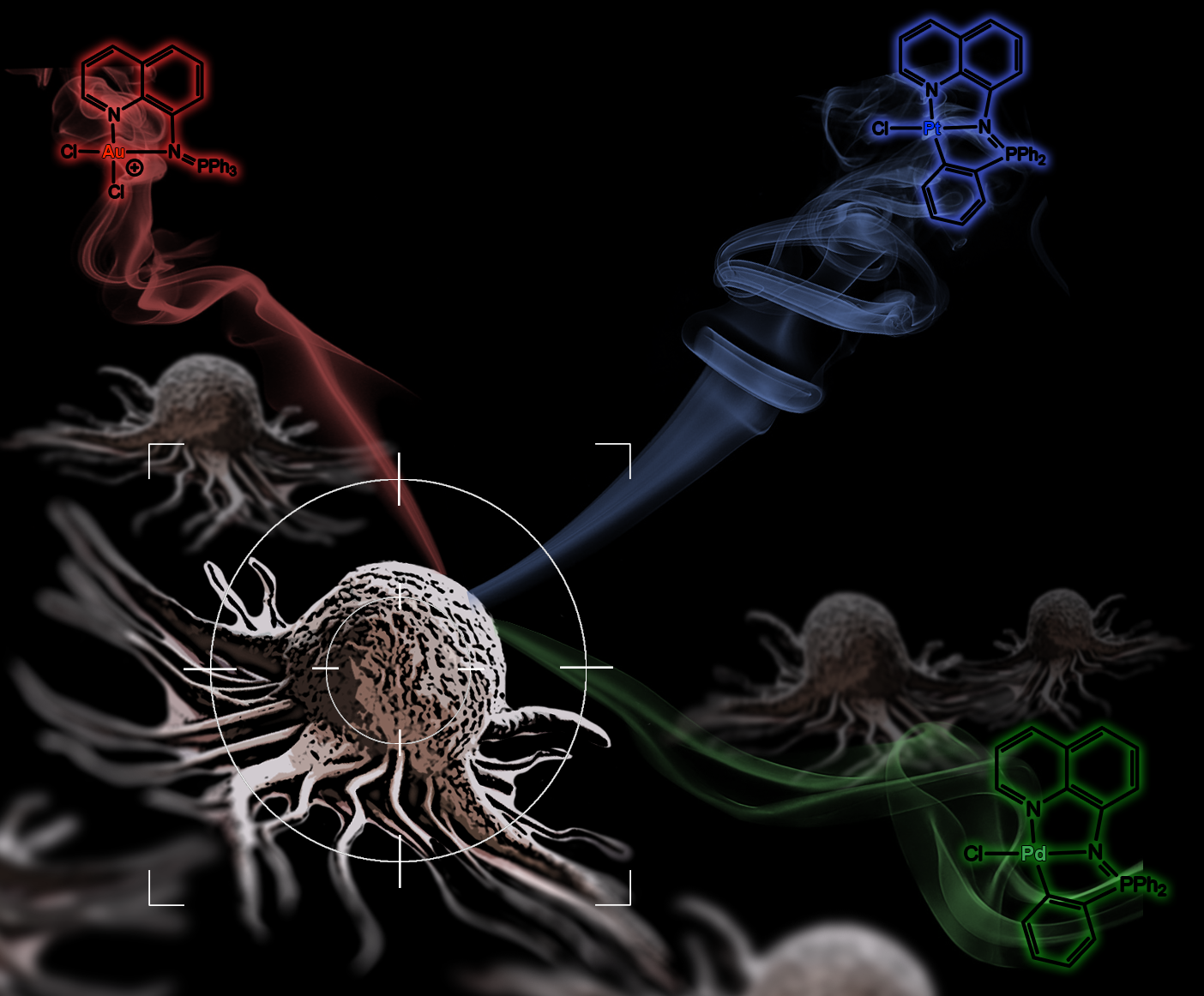
Metal-based drugs as anticancer agents
I am also interested in the mechanism of action of metal-based drugs as potential anti-cancer therapies. More specifically, my work focuses on metal-based drugs with fluorescence properties that can be used as probes or trackers for various cell functions and have anti-cancer properties.
Highlighted publications:
– de Almeida, A. & Bonsignore, R.; Fluorescent metal-based complexes as cancer probes. Bioinorg. & Med. Chem. Letters, (2020)
– Wragg, D., et al. On the mechanism of gold/NHC compounds binding to DNA G-quadruplexes: combined metadynamics and biophysical methods. Angew. Chem. Int. Ed. Engl., (2018)
Publications
For the most up-to-date information, please visit ORCID.
*authors contributed equally to the work; # corresponding author
2023
27 – (Pre-print) Srinivasan, S., Baskaran, S., Mukhopadhyay, S., Gamage, M.P., Ng, V., Dalavaye, N., de Almeida, A., Hassoulas, A.; Challenging Pedagogical Styles of Teaching: Amalgamating Blended Learning with Near-peer Teaching for Integrated Structured Clinical Examination (ISCE) Preparation.
26 – Hassoulas, A., de Almeida, A., West, H., Abdelrazek, M., Coffey, M.J.; Developing a personalised, evidence-based and inclusive (PEBIL) model of blended learning: a cross-sectional survey. Education and Information Technologies, (2023), DOI: 10.1007/s10639-023-11770-0
2021
25 – de Almeida, A.#, Parthimos, D., Dew, H., Smart, O., Wiltshire, M., Errington, R.J.; Aquaglyceroporin-3’s Expression and Cellular Localization Is Differentially Modulated by Hypoxia in Prostate Cancer Cell Lines. Cells, (2020) 10(4), 838; https://doi.org/10.3390/cells10040838
2020
24 – de Almeida, A., Bonsignore, R.; Fluorescent metal-based complexes as cancer probes. Bioinorg. & Med. Chem. Letters, (2020) 30(13), DOI: 10.1016/j.bmcl.2020.127219
2019
23 – Wragg, D., de Almeida, A., Casini, A. and Leoni, S.; Unveiling the mechanisms of aquaglyceroporin-3 water and glycerol permeation by metadynamics, Chem.: Eur. J, (2019) DOI: 10.1002/chem.201902121
22 – Wenzel, M.N., Mósca, A., Graziani, V., Aikman, B., Thomas, S., de Almeida, A; Insights into the Mechanisms of Aquaporin-3 Inhibition by Gold(III) Complexes: the Importance of Non-Coordinative Adduct Formation, Inorganic Chemistry, (2019) DOI: 10.1021/acs.inorgchem.8b03233
2018
21 – Aikman, B., Wenzel, M.N., Mósca, A., Klooster, W.T, Coles, S.J., de Almeida, A., Soveral, G., Casini, A.; Gold(III) pyridine-benzimidazole complexes as aquaglyceroporin inhibitors and antiproliferative agents. Inorganics, (2018); DOI: 10.3390/inorganics6040123
20 – de Almeida, A.*, Mósca, A.*, et al., Molecular basis of aquaporin-7 permeability regulation by pH; Cells, (2018), DOI: 10.3390/cells7110207
19 – de Almeida, A.*, Wragg, D.*, Bonsignore, R.*, Kuehn, F.E., Leoni, S., Casini, A.; On the mechanism of gold/NHC compounds binding to DNA G-quadruplexes: combined metadynamics and biophysical methods. Angew. Chem. Int. Ed. Engl., (2018), DOI: 10.1002/anie.201805727
18 – Aikman, B., de Almeida, A., Meier-Menches, S. M., Casini, A.; Aquaporins in cancer development: opportunities for bioinorganic chemistry to contribute novel chemical probes and therapeutic agents. Metallomics, (2018), DOI: 10.1039/C8MT00072G
2017
17 – Boorsma, C.E., Putri, K.S.S., de Almeida, A., et al., (2017) The Role of Tartrate Resistant Acid Phosphatase in Alveolar Macrophages in the Context of Obstructive Pulmonary Diseases, Sci. Rep. DOI: 10.1038/s41598-017-12623-w
16 – de Almeida, A.*, Mósca, A.*, et al.; The mechanism of aquaporin inhibition by gold compounds elucidated by biophysical and computational methods. Chem. Comm., (2017) DOI: 10.1039/ C7CC00318H
2016
15 – de Almeida, A.*, Khan, R.A.*, et al.; Transition-metal norharmane compounds as possible cytotoxic agents: New insights based on a coordination chemistry perspective. J. Inorg. Biochem., (2016) DOI: 10.1016/j.jinorgbio.2016.07.001
14 – de Almeida, A.*, Martins, A.P*, et al.; Exploring the gating mechanisms of aqua- porin-3: new clues for inhibitor’s design? Mol. Biosyst. (2016) DOI: 10.1039/C6MB00013D
13 – Tsitsa, I., Tarushi, A., Kalogiannis, S., Perdih, F., de Almeida, A., et al.; Structure and biological perspectives of metal complexes of flumequine. RSC Advances (2016) DOI: 10.1039/C5RA25776J
12 – de Almeida, A.*, Wenzel, M.*, et al.; New Luminescent Polynuclear Metal Complexes with Anticancer Properties: Toward Structure-Activity Relationships. Inorg. Chem., (2016), DOI: 10.1021/acs.inorgchem.5b02910
2015
11 – Book Chapter: Casini, A. and de Almeida, A. (2015) Aquaporins in health and disease: New molecular targets for drug discovery. Chapter: Inorganic compounds as aquaporin substrates or as potent inhibitors: a coordination chemistry point of view. CRC Press/Taylor & Francis Group Access here
10 – Spinello, A., de Almeida, A., Casini, A. and Barone, G.; The inhibition of glycerol permeation through aquaglyceroporin-3 induced by mercury(II): a molecular dynamics study. J.Inorg. Biochem., (2015), DOI:10.1016/j.jinorgbio.2015.11.027
2014
9 – de Almeida, A., Soveral, G., Casini, A.; Gold compounds as aquaporin inhibitors: new opportunities for therapy and imaging. Med. Chem. Comm., (2014) DOI: 10.1039/c4md00265b
8 – Khan, R.A, Al-Farhan, K., de Almeida, A., et al.; Light-stable bis(norharmane)silver(I) compounds: synthesis, characterization and antiproliferative effects in cancer cells. J. Inorg. Biochem., (2014) DOI: 10.1016/j.jinorg- bio.2014.06.019
7 – de Almeida, A.*, Madeira, A.*, et al.; A Gold Coordination Compound as a Chemical Probe to Unravel Aquaporin-7 Function. Chem. Bio. Chem., (2014) DOI: 10.1002/cbic.201402103
6 – de Almeida, A.*, Bertrand, B.*, et al.; New Gold(I) Organometallic Compounds with Biological Activity in Cancer Cells. Eur. J. Inorg. Chem., (2014) DOI: 10.1002/ejic.201402248(Front cover picture)
5 – Rubbiani, R., Salassa, L., de Almeida, A., Casini, A. and Ott, I.; Cytotoxic Gold(I) N-heterocyclic Carbene Complexes with Phosphane Ligands as Potent Enzyme Inhibitors. Chem. Med. Chem.; (2014) 9: 1205–1210. DOI: 10.1002/cmdc.201400056 (VIP paper, Front cover picture)
4 – Frik, M., Jiménez, J., Vasilevski, V., Carreira, M., de Almeida, A., Gascón, E., Benoit, F., Sanaú, M., Casini, A. and Contel, M.; Luminescent iminophosphorane gold, palladium and platinum complexes as potential anticancer agents. Inorg. Chem. Front., (2014) 1, 231-241 DOI: 10.1039/C4QI00003J (Front cover picture)
2013
3 – Lease, N., Vasilevski, V., Carreira, M., de Almeida, A., Sanaú, M., Hirva, P., Contel, M.; Potential anticancer heterometallic Fe-Au and Fe- Pd agents: initial mechanistic insights. J. Med. Chem., (2013) 56 (14), pp 5806–5818 DOI: 10.1021/jm4007615
2 – Martins, A. P., Ciancetta, A., de Almeida, A., Marrone, A., Re, N., Soveral, G. and Casini, A.; Aquaporin Inhibition by Gold(III) Compounds: New Insights. Chem. Med. Chem., (2013) 8: 1086–1092. DOI: 10.1002/cmdc.201300107
1 – de Almeida, A., Oliveira, B. L., Correia, J. D. G., Soveral, G., & Casini, A.; Emerging protein targets for metal-based pharmaceutical agents: An update. Coordination Chemistry Reviews, (2013) 257 (19-20) 2689-2704. DOI:10.1016/j.ccr.2013.01.031